SIGA announced Health Canada regulatory approval of Oral TPOXX
On Dec. 1, 2021, SIGA Technologies announced that Health Canada had approved oral TPOXX (tecovirimat) as an extraordinary…
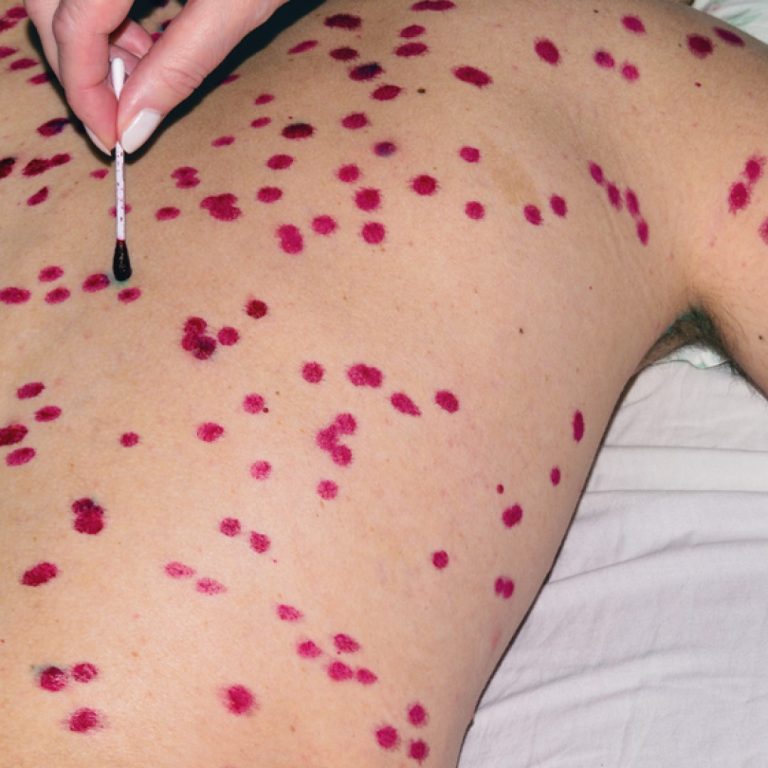
On Dec. 1, 2021, SIGA Technologies announced that Health Canada had approved oral TPOXX (tecovirimat) as an extraordinary…
On Nov. 30, 2021, Inovio Pharma announced the company was rapidly moving to evaluate its COVID-19 DNA vaccine…
The COVID-19 Roller Coaster is a wild ride, strap yourself in and hold on… Cast of Characters: Senator…

On Nov. 26, 2021, Merck provided an update on the MOVe-OUT study of molnupiravir, an investigational oral antiviral…
On Nov. 24, 2021, Novavax announced its submission to the Singapore Health Sciences Authority for interim authorization of…

On Nov. 24, 2021, Johnson & Johnson announced the U.S. Food and Drug Administration (FDA) had issued Emergency…
On Nov. 22, 2021, Tonix Pharmaceuticals announced the publication of ‘Sangivamycin is highly effective against SARS-CoV-2 in vitro…
On Nov. 19, 2021, the U.S. Food and Drug Administration (FDA) announced that it had extended the emergency…
On Nov. 18, 2021, Pfizer announced an agreement with the U.S. government to supply 10 million treatment courses…

On Nov. 17, 2021, Novavax and and Serum Institute of India announced that the Philippine Food and Drug…

On Nov. 17, 2021, Novavax announced that the European Medicines Agency (EMA) had begun its evaluation of an…

On Nov. 17, 2021, Zosano Pharma announced that the Philippine Food and Drug Administration had granted emergency use…
On Nov. 17, 2021, Pacific Northwest National Laboratory (PNNL) announced researchers had compiled the most comprehensive road map…

On Nov. 17, 2021, GlaxoSmithKline and Vir Biotechnology announced U.S. government contracts totalling approximately $1 billion (USD) to…

On Nov. 16, 2021, Pfizer announced it had submitted an Emergency Use Authorization (EUA) of its investigational oral…

On Nov. 16, 2021, an agreement was announced that enabled the European Union and European Economic Area countries…

On Nov. 15, 2021, Novavax and SK bioscience announced submission of a Biologics License Application (BLA) for Novavax’…
On Nov. 15, 2021, Moderna confirmed that Health Canada had authorized the use of a booster dose of…

On Nov. 15, 2021, AstraZeneca reported that 2 billion doses of the COVID-19 vaccine had been released for…
On Nov. 12, 2021, GlaxoSmithKline and Vir Biotechnology announced headline data from the randomised, multi-centre, open-label COMET-TAIL Phase…

On Nov. 10, 2021, the World Health Organization (WHO) announced that while reported measles cases had fallen compared…

On Nov. 9, 2021, Moderna announced that it has submitted for a variation to the conditional marketing authorization…

On Nov. 9, 2021, Inovio Pharma announced that the U.S. Food and Drug Administration (FDA) provided authorization to…
On Nov. 8, 2021, Regeneron announced that the European Commission had approved the casirivimab and imdevimab antibody cocktail,…

On Nov. 8, 2021, Cocrystal Pharma announced that its SARS-CoV-2 main protease inhibitors showed potent in vitro pan-viral…

On Nov. 5, 2021, the USDA’s (USDA) National Veterinary Services Laboratories announced confirmation of SARS-CoV-2 in two spotted…

On Nov. 5, 2021, Pfizer announced it was investigational novel COVID-19 oral antiviral candidate, PAXLOVID, significantly reduced hospitalization…

On Nov. 5, 2021, Chugai Pharmaceutical, announced that it had obtained approval from the Ministry of Health, Labour…

On Nov. 3, 2021, Novavax announced the company had filed for provisional approval of the vaccine to the…

On Nov. 3, 2021, Inovio Pharma announced that it had received authorization from India’s Central Drugs Standard Control…