FDA approved AstraZeneca’s Imfinzi for limited-stage small cell lung cancer
On Dec. 4, 2024, The U.S. Food and Drug Administration (FDA) announced approved of AstraZeneca’s blockbuster drug Imfinzi…
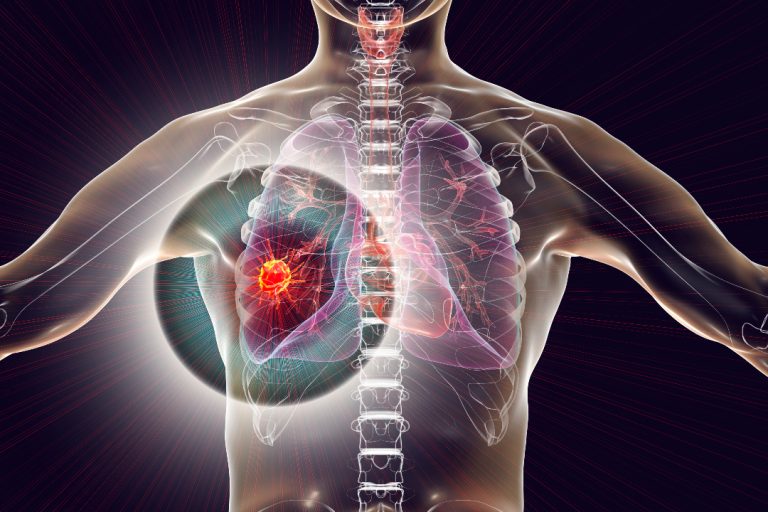
On Dec. 4, 2024, The U.S. Food and Drug Administration (FDA) announced approved of AstraZeneca’s blockbuster drug Imfinzi…
On Nov. 21, 2024, the U.S. Centers for Disease Control and Prevention (CDC) reported that by the week…

On Nov. 15, 2024, the White House Office of Science and Technology Policy released a report on Building…

On Nov. 14, 2024, the U.S. Food and Drug Administration approved PTC Therapeutics’ Kebilidi (eladocagene exuparvovec-tneq), an adeno-associated…

On Nov. 13, 2024, the U.S. Food and Drug Administration (FDA) and the U.S. Centers for Disease Control…
On Nov. 4, 2024, the U.S. Food and Drug Administration (FDA), in collaboration with the Environmental Protection Agency…

On Oct. 28, 2024, HOPO Therapeutics announced that it has been awarded a contract valued at up to…
On Oct. 22, 2024, Pfizer announced that the U.S. Food and Drug Administration (FDA) had approved ABRYSVO® (Respiratory…

On Oct. 16, 2024, The U.S. Food and Drug Administration (FDA) announced it had updated the label for…

On Oct. 14, 2024, RedHill Biopharma announced that the U.S. government’s Biomedical Advanced Research and Development Authority (BARDA)…
On Oct. 11, 2024, Pfizer announced that the U.S. Food and Drug Administration (FDA) had approved HYMPAVZI™ (marstacimab-hncq)…
On Oct. 10, 2024, Genentech announced today that the U.S. Food and Drug Administration (FDA) approved ItovebiTM (inavolisib),…

On Oct. 8, 2024, Okanagan Specialty Fruits® (OSF), the developer and grower behind the innovative Arctic® apple varieties, announced that its…

On Oct. 4, 2024, Exact Sciences announced the U.S. Food and Drug Administration (FDA) had approved the Cologuard…

On Oct. 2, 2024, a study from the GBD Tobacco Forecasting Collaborators reported that by accelerating the decline…

On Oct. 1, 2024, the U.S. Food and Drug Administration (FDA) announced that the unified Human Foods Program,…

On Sept. 30, 2024, Amneal Pharmaceuticals announced that it had launched CREXONT ® (carbidopa and levodopa) extended-release capsules…

On Sept. 26, 2024, the U.S. Food and Drug Administration (FDA) approved Bristol-Myers Squibb’s Cobenfy (xanomeline and trospium…

On Sept. 20, 2024, the U.S. Food and Drug Administration approved FluMist for self- or caregiver-administration. FluMist is…

On Sept. 13, 2024, Genentech announced that the U.S. Food and Drug Administration (FDA) had approved Ocrevus Zunovo™…

On Sep. 12, 2024, the the U.S. Food and Drug Administration (FDA) authorized the first over-the-counter (OTC) hearing…

On Aug. 30, 2024, Novavax announced the Novavax COVID-19 Vaccine, Adjuvanted (2024-2025 Formula) (NVX-CoV2705) had received Emergency Use…

On Aug. 7, 2024, Amneal Pharmaceuticals announced that the U.S. Food and Drug Administration (FDA) had approved CREXONT…
On Aug. 6, 2024, the U.S. Food and Drug Administration (FDA) announced it had approved vorasidenib (Voranigo, Servier…

On Aug. 2, 2024, the U.S. Food and Drug Administration approved Adaptimmune’s Tecelra (afamitresgene autoleucel), a gene therapy…
On Jun. 27, 2024, the U.S. Food and Drug Administration granted marketing authorization to Cepheid for the Xpert…

On Jun. 20, 2024, the U.S. Food and Drug Administration expanded the approval of Sarepta Therapeutics’ Elevidys (delandistrogene…

On Jun. 17, 2024, Merck announced that the U.S. Food and Drug Administration (FDA) had approved CAPVAXIVE™ (Pneumococcal…
On Jun. 14, 2024, the FDA’s Vaccines and Related Biological Products Advisory Committee (VRBPAC) unanimously voted to recommend…

On Jun. 10, 2024, Roche announced that the U.S. Food and Drug Administration (FDA) had granted Emergency Use…